After scientific breakthrough: Digid seeks FDA approval for its rapid corona antigen test
At the Helmholtz Centre for Infection Research (HZI), scientists have succeeded for the first time in detecting SARS-CoV-2 viruses using the revolutionary Cantisense™ technology from Digital Diagnostics AG. Application for approval of the Digid Cantisense™ SARS-CoV-2 Test by the FDA has been submitted. With the new digid Cantisense™ SARS-CoV-2 test, corona infections can be reliably detected within minutes at the point-of-care. Delivery of the first point-of-care tests is planned for July.
At the high-security laboratory of the Helmholtz Centre for Infection Research (HZI) in Braunschweig, Germany, the revolutionary Cantisense™ technology of the German health technology company Digital Diagnostics AG was used for the first time to successfully detect SARS-CoV-2 viruses in high measuring accuracy in test liquids.
In contrast to PCR testing, the new test provides a clear electronic “YES” or “NO” information within a few minutes, saving precious time in the diagnosis. What’s more, the Digid Cantisense™ SARS-CoV-2 Test directly detects the presence of the virus while other rapid tests only recognize antibodies. Antibodies are formed when a patient was already infected and contagious for several days. Therefore, rapid antibody tests are only of limited use to contain the spread of the pandemic.
Konstantin Kloppstech, CTO at Digital Diagnostics, says: “Recent test series at the HZI high-security laboratory have shown that SARS-CoV-2 viruses can be detected directly and reliably using our Cantisense technology and without the need for PCR or further sample processing. This is a scientific breakthrough. We have coated cantilevers with a capture layer of highly specific monoclonal antibodies, which can reliably bind SARS-CO-2 viruses in the test fluid.”
Constantin von Gersdorff, CEO of Digital Diagnostics, said: “The next step will be to initiate clinical studies with patient samples. To this end, we have already established international collaborations with leading hospitals in Europe, Africa and Asia”.
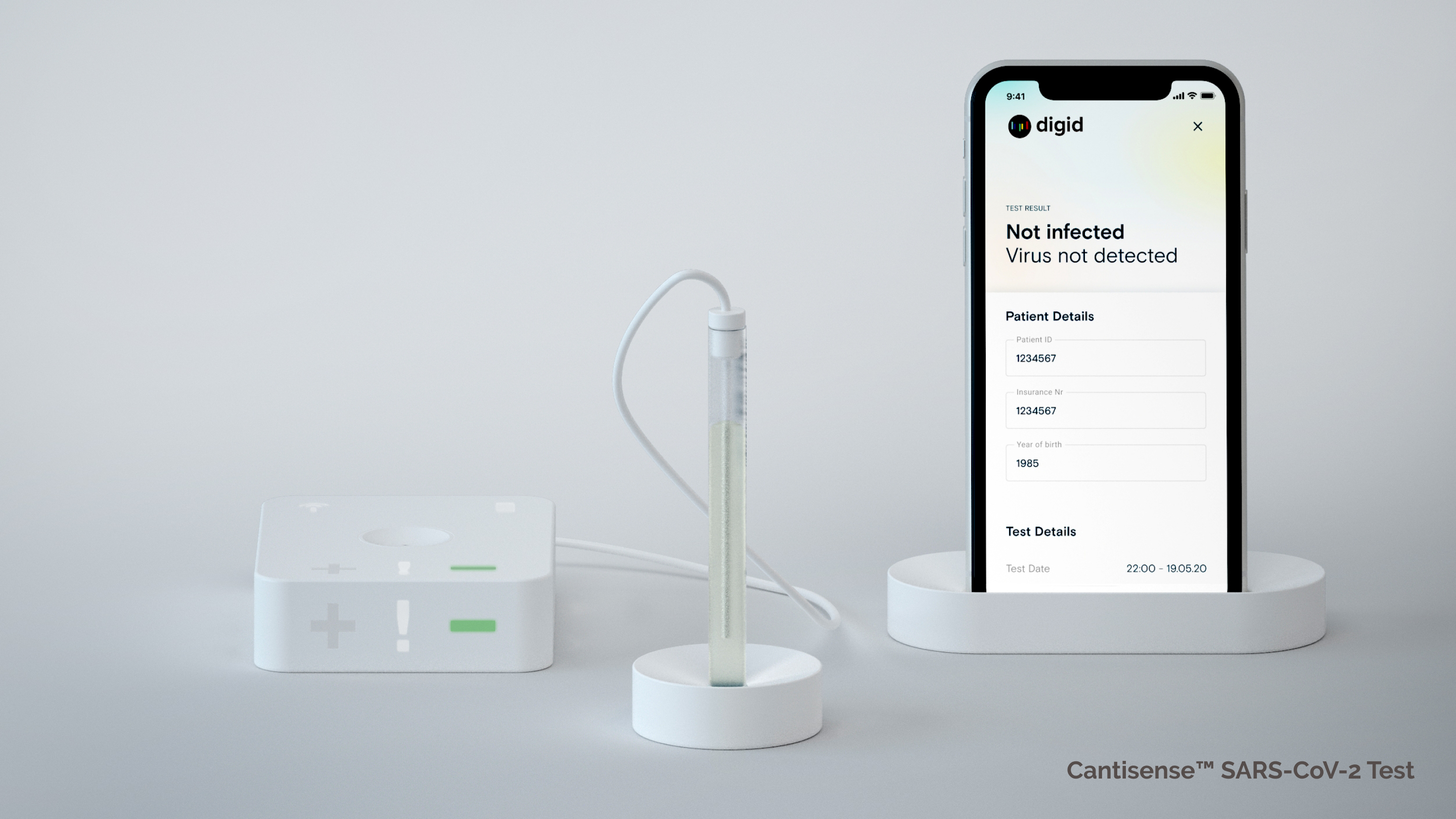
Point-of-care test kit: sensor hub, biosensor, mobile device (prototype/copyright: Digital Diagnostics AG)
With digid´s point-of-care solution, the measurement results are transmitted wirelessly from a sensor hub to a mobile device. By connecting to a secure analytics platform, the numerous sensor data can be augmented with further data and processed anonymously for research and diagnostic applications.
Due to its measurement speed and the highly reliable results, the Digid Cantisense™ SARS-CoV-2 Test is particularly suitable to support the containment of the current SARS-CoV-2 pandemic. The test offers the possibility of simple and rapid testing of patients and medical staff as an alternative to laboratory tests and enables reliable identification of infected persons within minutes. The possible areas of application therefore also include screening for access control at airports and railway stations, hospitals and specially protected areas (such as retirement and nursing homes) as well as for companies who want to ensure that their production runs smoothly and safely.
Digital Diagnostics has applied for approval of the Digid Cantisense™ SARS-CoV-2 Test by the US Food and Drug Administration (FDA). Digid aims to provide millions of units by July 2020. A further scaling of capacities is planned in the short term. The experts at Digital Diagnostics are also working at full speed to further develop the technology and to adapt the point-of-care testing procedure for use by consumers at home.
Partner Konsortium
digid-Digital Diagnostics AG, Mainz, Berlin
Helmholtz-Zentrum für Infektionsforschung (HZI), Braunschweig
Laboratories for Natural Product and Conjugation Chemistry (naconLabs)
Abteilung Mikrorobotik und Regelungstechnik (AMiR) an der Universität Oldenburg
Centre for Biosensors, Bioelectronics and Biodevices, University of Bath
Łukasiewicz-Instytut Technologii Elektronowej, Warschau
Press contact Consortium
Thomas Huber
semanticom GmbH
+49 151 14 96 58 10
digid-pr@semanticom.eu





